
Transformation of QMS: why evolution is inevitable and urgent
10 min read 23 February 2026
The pharmaceutical industry is experiencing a rapid acceleration in the adoption of artificial intelligence (AI), automation and advanced digital technologies across R&D. These capabilities are already reshaping how data is analysed, decisions are made and development timelines are compressed. AI is no longer an emerging concept it is becoming embedded in everyday ways of working.
However, while technology is evolving at pace, the processes it operates within and the way they are governed through Quality Management Systems (QMS) often remain anchored in pre-digital assumptions. Many QMS frameworks were designed for linear, manual, document-centric environments, relying heavily on static SOPs and human oversight. When advanced digital tools are layered onto these legacy processes, their ability to deliver value at scale is limited.
To realise the full potential of AI, organisations must evolve not only their technology but their end-to-end processes along with how those processes are managed and assured. This shift is unavoidable. As more tasks and decisions are executed by systems rather than individuals, quality must be built into workflows from the outset and continuously monitored across interconnected platforms. Organisations that embrace this evolution will be best positioned to unlock the benefits of AI while maintaining regulatory confidence, operational resilience and patient safety.
AI in R&D: speed and insight redefined
AI is unlocking new frontiers in pharmaceutical research and development. AI algorithms can analyse complex data faster than ever before, optimise clinical trial design and accelerate decision-making at every stage of the pipeline. These capabilities are no longer speculative, regulators now acknowledge the transformative role of AI in drug development with an increasing focus on quality assessment and assurance. In early 2025, the FDA issued new draft guidance providing a risk-based framework for assessing AI models used to support regulatory decision-making about drug safety, effectiveness, and quality underscoring AI’s growing role in regulated product lifecycles.
As organisations accelerate the development, deployment, and adoption of AI across R&D to sustain competitive advantage, traditional quality frameworks are coming under increasing strain. The promise of compressed development timelines and expanded AI-enabled capabilities is exposing the limitations of quality models designed for predominantly manual ways of working.
The inevitability of QMS transformation – moving beyond SOPs to a scalable process architecture
At its core, quality management ensures that processes are controlled, compliant, and capable of delivering products that are safe and effective. Historically, this has involved high-volumes of Standard Operating Procedures (SOPs), extensive manual documentation and risk controls centred on human oversight.
This paradigm is under pressure to evolve; interconnected systems, digital platforms and AI-enabled analytics are reshaping how quality work is done.
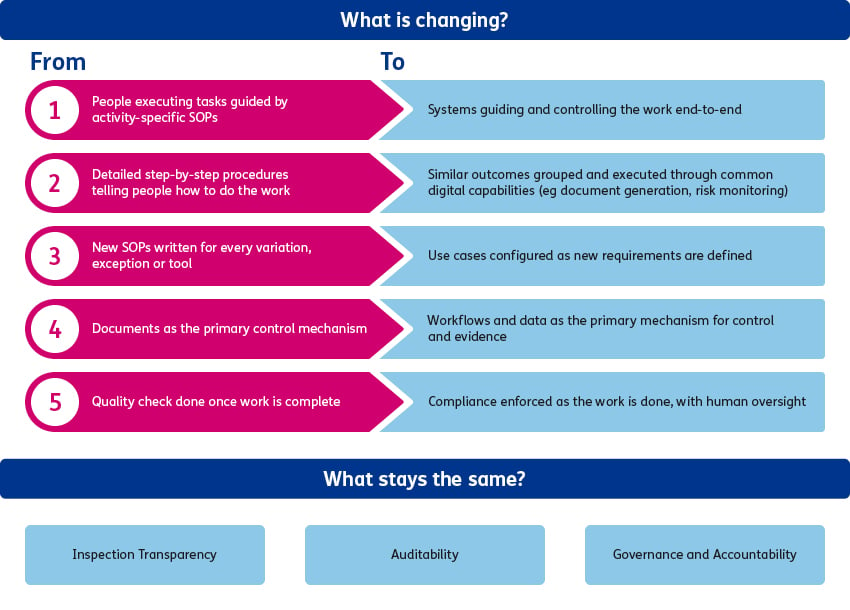
The future QMS is built on a simple but powerful principle: separate what must remain stable from what needs to adapt. Processes that deliver similar outcomes are designed around a small number of regulatory defensible core steps that rarely change. A stable process archetype defines the key activities to be executed and provides the consistency, auditability and inspection confidence.
Within a process archetype, execution of the activity can vary safely through configuration rather than new procedures. Context-specific metadata, rules, data sources, roles, and digital tools are adjusted to support different use cases, while the underlying process steps remain.
A simple example is document generation. Instead of creating new procedures for each document type, organisations use a single, stable document-generation process that defines the required quality steps. Different document outputs such as protocols, CSRs, or informed consent forms are produced by configuring data inputs, business rules, reviewer roles, and approval requirements within that backbone. The underlying process remains validated and inspection-ready, while additional use cases and changes in ways of working can be adapted over time through controlled workflow configuration rather than repeated procedural updates.
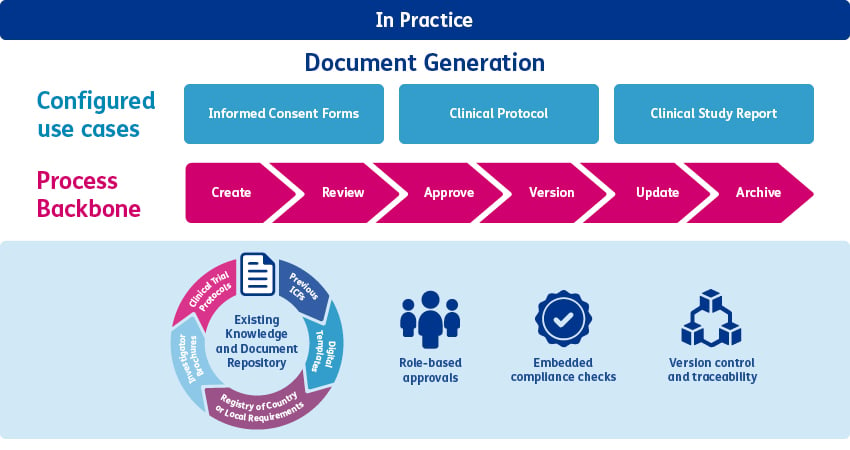
Embedded compliance: turning quality into a digital workflow
In a digital QMS ecosystem:
- Compliance is not an afterthought checked manually it becomes embedded into automated workflows.
- Deviations, CAPAs, change controls and audits can be triggered, tracked, and closed with real-time traceability.
- Predictive analytics can anticipate issues before they become business-critical events, shifting quality control from reactive to proactive.
This transformation yields clear benefits:
- Reduced human error
- Faster, more accurate and up to date documentation
- Greater transparency for audits and inspections
- Enhanced risk identification and mitigation
It is important to remember however; automation does not eliminate the need for quality oversight it changes what quality professionals oversee.
Organisational considerations: are we ready?
Moving to embedding quality into digital workflows brings with it organisational implications:
- QMS is not reduced-it is reimagined
The shift away from paper-intensive SOPs does not diminish quality requirements; it reframes them. Instead of managing a sprawling procedural library, organisations must now manage digital process design, automated decision points and software lifecycle assurance.
- Validation, Qualification, and Computerised System Assurance (CSA)
With more tasks executed by systems, the emphasis on Computer Systems Assurance and continuous qualification intensifies. AI models themselves may need to be subjected to lifecycle validation, risk assessment, version control and performance in line with GxP process requirements.
- Skills and Quality Expertise
Traditional quality professionals excel in compliance, risk management and SOP governance but digital quality demands different competencies such as data literacy, software validation expertise, AI governance acumen and an understanding of automated systems dynamics. Organisations must assess whether current quality teams have the necessary skills and, if not, develop robust upskilling or recruitment strategies.
Generative AI: a special challenge
Generative AI, tools that produce text, insights and recommendations offer significant opportunities to accelerate procedures, reports, and regulatory documentation. However, limited explainability and reproducibility introduce risks when outputs influence GxP-regulated activities.
For QMS this creates new challenges: organisations must define accountability, ensure traceability and demonstrate that AI-driven workflows operate within validated boundaries. Traditional validation approaches may be insufficient, requiring risk-based oversight, model lifecycle management and continuous monitoring.
Recent EMA guidance on the use of Large Language Models (LLMs) highlights the need for human oversight, transparency, and control, signalling that QMS must evolve to govern AI-enabled processes. Without such alignment, generative AI may become a compliance bottleneck rather than an accelerator, particularly where auditability and defensible decision-making are critical to product quality and patient safety.
A balanced path forward: strategy and governance
To responsibly embrace AI and begin their digital QMS transformation, organisations need to consider:
- Developing a digital quality strategy
Align technology deployment with business goals, regulatory expectations and quality culture. Avoid point solutions in isolation.
- Building a Governance Framework
Ensure AI models, workflows, and automated processes are governed with clear ownership, risk controls and continuous monitoring.
- Defining capability-based process archetypes
Identify opportunities to consolidate related activities, such as document generation or data processing under adaptable centralised digital processes. This ensures consistency, traceability, and compliance while enabling scalable, AI-enabled operations.
- Reframing quality roles
Transition quality teams from gatekeepers of SOP compliance to architects of digital quality ecosystems. Review your workforce plan to ensure you have the right skillsets to enable your QMS transformation.
- Engaging regulators early
Regulatory bodies like FDA and EMA encourage early engagement on AI applications that influence regulatory decisions particularly those affecting product quality and patient safety.
Conclusion: Digital quality is quality evolved
The digital transformation of Quality Management Systems is not a futuristic ideal, it is a strategic necessity. The convergence of AI, automation, and interconnected workflows promises significant gains in efficiency, compliance confidence, and product quality. Yet, realising these gains requires organisational maturity, new capabilities, and a willingness to rethink entrenched practices.
Pharmaceutical organisations that proactively embrace digital QMS with thoughtful governance, regulatory alignment and a focus on people as much as technology will be best positioned to capitalise on the opportunities AI and advanced technologies unlock whilst successfully navigating the complexities of their regulated use.
Evolution of QMS isn’t just preferable; in the digital age it is unavoidable.
Our Experts
Related Insights

AI in healthcare needs more than hype – it needs quality
Digital and AI innovation is pushing healthcare and life sciences into a new era, creating fresh opportunities to unlock value and improve patient outcomes.
Read more
Reimagining a critical R&D process through AI
A top 10 global biopharmaceutical company partnered with Baringa to address a challenge familiar across R&D.
Read more
Enabling commercial agility in medical device manufacturing
A global medical device company was operating with limited visibility across its commercial and operational functions.
Read more
A focused integrated evidence planning approach that identified over >£10m of savings in one year
Our client was a leading global pharmaceutical company with an ambitious 5-year pipeline of launches that required a cross-functional step-change in how they approached evidence generation.
Read moreIs digital and AI delivering what your business needs?
Digital and AI can solve your toughest challenges and elevate your business performance. But success isn’t always straightforward. Where can you unlock opportunity? And what does it take to set the foundation for lasting success?
